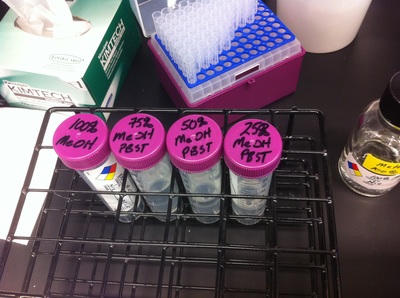
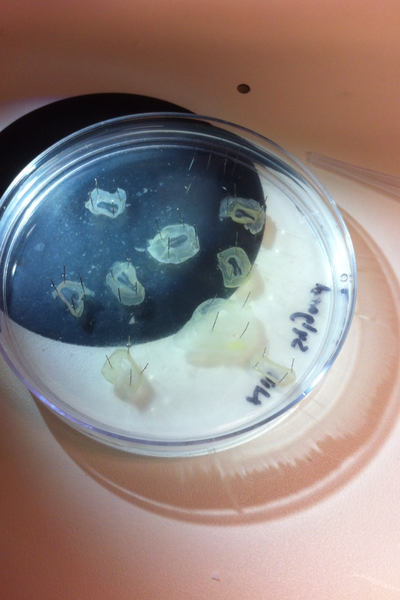
After several weeks of cracking open dozens and dozens of eggs, finally, the day has come. On Friday, October 2nd, we were able to successfully extract 11 chick embryos into a petri dish full of ringer solution. Afterwards, we separated the embryos from the paper slides by gently dropping ringer solution around the slide while using forceps. We then proceeded to fix these embryos by crucifying them: pinning each embryo on the corners with insect pins on a silkgard plate full of ringer solution. Pinning the embryos took quite some time, especially because the embryos tended to fold into themselves often. We would use the ringer solution and pipette around the embryos to spread them out fully. We used forceps to place pins on each corner of the embryo. After successfully pinning all of the embryos in the silkgard plate, we removed the ringer solution and replaced it with 4% paraformaldehyde in order to fix the embryos. On Tuesday, October 6th, we replaced the paraformaldehyde with PTW. We did this because antigens are often sensitive to over-fixing and all PFA must be washed out. On Wednesday, October 7th, Toby & I performed three PBS with 0.1% Tween-20 washes with five minute intervals. Afterwards, we performed a series of Methanol washes to dehydrate the embryos to help penetration. We washed them once with 25% MeOH, once with 50% MeOH, once with 75% MeOH, and three times with 100% MeOH. We then left the vial of chick embryos in 100% MeOH overnight at -20 degrees Celsius. During the five minute intervals between each wash, we autoclaved glassware by placing aluminum foil and autoclave tape on the tops of the glassware and placing them in the autoclave machine under the instrumentation program. On Thursday, October 8th, we rehydrate the chick embryos through the same methanol and PBS with 0.1% Tween-20 washes.




 RSS Feed
RSS Feed
