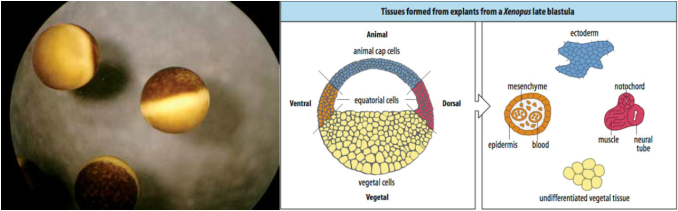
The embryonic development of an organism is one of the most essential that it will experience throughout its lifetime, with this period establishing the basic body plan and tissue types. The stages of early embryonic development in Xenopus laevis can be summarized by: cleavage, blastulation, gastrulation, and neurulation. Gastrulation is arguably the most important, as it is during this stage that the three basic germ layers (ectoderm, mesoderm, and endoderm) along with the primary axes of the embryo are established to guide the differentiation of cells. However, even before gastrulation, the precursory cells of the embryo have already had their general fate decided. This is largely due to the presence of cytoplasmic determinants and other transcription factors that are distributed unevenly during the initial cleavage and blastulation. Although every cell contains the same genetic information, the differential gene expression that allows for cell and tissue differentiations within the embryo are driven by these early transcription factors. As a result, even during the blastula stages within X. laevis before the major determination and cell movement within gastrulation, the cells are already divided into regions that will be destined to become certain structures.
Because of this, it is possible to observe this precursory differentiation within key regions of the developing embryo, one of which being the animal cap at the most anterior part of the animal pole. In the lab this week, I attempted to dissect this region of the embryo and study what would result of this extracted tissue.
Because of this, it is possible to observe this precursory differentiation within key regions of the developing embryo, one of which being the animal cap at the most anterior part of the animal pole. In the lab this week, I attempted to dissect this region of the embryo and study what would result of this extracted tissue.
In order to perform this procedure, the general steps are this: procure eggs in the late blastula phase (approx. stage 8.5 - 9), physically remove the vitelline membrane surrounding the embryo with a pair of forceps, carefully snip out a small square of tissue from the animal pole using one forcep with the other holding the embryo in place, and place this tissue from the animal pole within a solution of 0.5M MMR to develop. Shortly after the tissue is extracted from the main embryo, it will mitotically divide to produce a small sphere of what would become ectodermal tissue. If the tissue was contaminated with precursory mesodermal tissue as the animal cap was dissected, the resulting sphere would have a more oblong and amorphous shape.
This dissected tissue could then be further studied through the injection of RNA, growth factors, etc. in order to induce further developments within the tissue. My initial experience with animal cap dissections has reinforced the notion that cells are already aligned on a certain path of development even before gastrulation, and has revealed to me the amazing capacity of embryonic tissues to regenerate despite mortification and act as useful models for other developmental processes.
References:
Hollemann TC, Yonglong; Sölter, Marion; Kühl, Michael; Pieler, Thomas. 2006. Embryonic explants from Xenopus laevis as an assay system to study differentiation of multipotent precursor cells. Pages 191-198 in Celis JE, ed. Cell Biology: A Laboratory Handbook, vol. 1 Elsevier Inc.
Images:
http://www.awionline.org/pubs/Quarterly/08-57-03/08_57_3p25.htm
http://www.mun.ca/biology/desmid/brian/BIOL3530/DB_03/fig3_50.jpg



 RSS Feed
RSS Feed
