According to the Central Dogma of Genetics, following transcription, mRNA is translated into protein to become functional within the cell. If the process of translation were inhibited for a certain gene, the function of that gene would be lost. One such method of performing this is injection with a morpholino, an oligonucleotide which has the capability to bind to specific mRNA transcripts within a live embryo and physically inhibit translation by blocking the mRNA from binding to the ribosome. Being injected into a specific cell during an early stage of Xenopus laevis development, the loss-of-function effect of the Morpholino can be localized to a general location based upon the cells that are progeny to the original injected cell. These experiments are performed with the intention of studying the migration of neural crest cells (observed through in situ hybridization with a sox10 probe) after injection with a morpholino targeting MMP-14 transcripts.
Hypothesis:
Embryos injected with a morpholino causing loss-of-function in regards to the expression of MMP-14 will show a decreased migration of neural crest cells distance down the dorsal-ventral axis and failure of segregation of migratory streams corresponding to rhombomeres of the hindbrain.
Experimental Design:
Xenopus laevis embryos are collected and fertilized in vitro. Upon reaching a four-cell stage, embryos are injected in one of the two cells of the animal pole that is fated to become the dorsal region of the embryo, these cells are smaller and lighter in color in comparison to the remaining two cells of the animal pole that are fated to develop into the ventral region. Two concentrations of morpholino are tested within current trials: 20ng and 50ng received per embryo. Along with the morpholino, embryos are injected with beta-galactosidase, which acts as a lineage tracer, indicating successful injection and demonstrating the general region in which the morpholino’s effect will be present. Injections were targeted towards the anterior-dorsal region of the embryo to affect the migratory streams of neural crest cells. Staining for beta-galactosidase (magenta specks seen in Injected samples) is performed after fixation of the embryos. Given the bilateral symmetry present in the embryo, one half of the embryo (the uninjected) acts as a control, while the injected may be compared against it, thus eliminating the confounding variable of individual variation in migration between embryos. After reaching the desired stage of 24 (in which the migratory streams of neural crest cells are very distinct), the embryos received staining for sox10 expression through in situ hybridization.
Results:
Hypothesis:
Embryos injected with a morpholino causing loss-of-function in regards to the expression of MMP-14 will show a decreased migration of neural crest cells distance down the dorsal-ventral axis and failure of segregation of migratory streams corresponding to rhombomeres of the hindbrain.
Experimental Design:
Xenopus laevis embryos are collected and fertilized in vitro. Upon reaching a four-cell stage, embryos are injected in one of the two cells of the animal pole that is fated to become the dorsal region of the embryo, these cells are smaller and lighter in color in comparison to the remaining two cells of the animal pole that are fated to develop into the ventral region. Two concentrations of morpholino are tested within current trials: 20ng and 50ng received per embryo. Along with the morpholino, embryos are injected with beta-galactosidase, which acts as a lineage tracer, indicating successful injection and demonstrating the general region in which the morpholino’s effect will be present. Injections were targeted towards the anterior-dorsal region of the embryo to affect the migratory streams of neural crest cells. Staining for beta-galactosidase (magenta specks seen in Injected samples) is performed after fixation of the embryos. Given the bilateral symmetry present in the embryo, one half of the embryo (the uninjected) acts as a control, while the injected may be compared against it, thus eliminating the confounding variable of individual variation in migration between embryos. After reaching the desired stage of 24 (in which the migratory streams of neural crest cells are very distinct), the embryos received staining for sox10 expression through in situ hybridization.
Results:
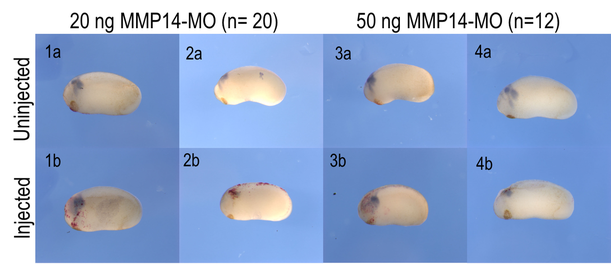
Figure 1. Embryos were injected with MMP14-MO and lineage tracer β-galactosidase in one of the dorsal animal cells. In situ hybridization was performed at stage 24 with sox10 antisense probe to determine the effect on neural crest migration. Upper panels (1a-4a) are lateral views of uninjected side and bottom panels (1b-4b) are lateral views of the injected side of the same embryos. Neural crest migrate was affected by MMP14-MO moderately or severely in different embryos. In embryos treated with 20 ng MO, 35% were unaffected, 45% were moderately affected, and 20% were severely affected. In embryos injected with 50 ng MO, 25% were unaffected, 58% were moderately affected, and 17% were severely affected. Perspective of images may skew observed results.
Discussion:
Injection with MMP-14 morpholino was found to produce moderate effects through decreased migration of neural crest cells at our tested concentrations in terms of migratory distance and segregation of migratory streams. Within the samples injected with 20ng, 65% of embryos were affected either moderately or severely, and within the samples injected with 50ng, 75% were affected either moderately or severely. These results indicate that loss-of-function through blocking translation of the MMP-14 gene does negatively impact the migration of neural crest cells at this stage of development.
Injection with MMP-14 morpholino was found to produce moderate effects through decreased migration of neural crest cells at our tested concentrations in terms of migratory distance and segregation of migratory streams. Within the samples injected with 20ng, 65% of embryos were affected either moderately or severely, and within the samples injected with 50ng, 75% were affected either moderately or severely. These results indicate that loss-of-function through blocking translation of the MMP-14 gene does negatively impact the migration of neural crest cells at this stage of development.
 RSS Feed
RSS Feed
