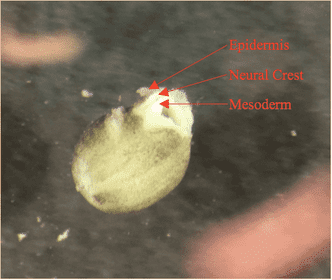
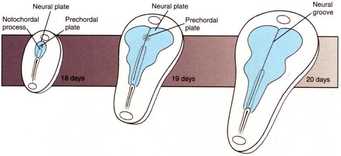
Neural crest tissue, being derived from ectoderm during neurulation and folding beneath the epidermis with the closing of the neural folds, is sandwiched between epidermis and mesoderm as the cells begin their migration. At stage 14 in development, as seen in the diagram above, the neural crest tissue occupies a tear drop-shaped area along the dorsal axis of the embryo. The migratory capacity of this tissue is valuable to our research of the MMP-14 gene, as matrix metalloproteinases are vital in digesting the ECM to allow for proper mesenchymal movement. However, secondary characteristics and roles of the MMP-14 protein, excluding its digestive properties, may be equally important to neural crest migration. As a result, this experiment was conducted to investigate the behavior of neural crest tissues that have been extracted and grown on a plate of fibronectin, with different treatments applied to knock-out MMP-14 expression. Fibronectin is a glycoprotein typically found in the extracellular matrix that allows for cell attachments, and was plated on petri dishes to allow for our neural crest explant to fuse to a stable surface and migrate. The rationale for observing explants outside of the embryo is that, fused to a simple layer of fibronectin, the dense and complex extracellular matrix that is normally present in the full embryo will be absent. Thus, the primary role of the MMP-14 protein in digesting this matrix will be unnecessary, and will isolate and allow for the exploration of the secondary roles of the protein in neural crest migration.
Hypothesis:
Neural crest explants in which the expression of MMP-14 has been knocked-out (either pre-translationally through morpholino or post-translationally through chemical inhibition) should demonstrate a reduced migratory distance and proper EMT of cells. This result is expected given previous results of whole mount in-situ hybridization with MMP-14 knock-outs. Although this experiment is essentially testing for inhibition of only the secondary roles of MMP-14, a decreased migratory capacity is still expected given MMP-14’s roles in signal transduction and activation of MMP-2.
Neural crest explants in which the expression of MMP-14 has been knocked-out (either pre-translationally through morpholino or post-translationally through chemical inhibition) should demonstrate a reduced migratory distance and proper EMT of cells. This result is expected given previous results of whole mount in-situ hybridization with MMP-14 knock-outs. Although this experiment is essentially testing for inhibition of only the secondary roles of MMP-14, a decreased migratory capacity is still expected given MMP-14’s roles in signal transduction and activation of MMP-2.
Experimental Design:
The protocol for dissection of the explants is based upon the methods provided in Borchers et al. 2000. The embryos used for dissection are between the stages of 12.5 and 14, during early neurulation when neural crest cells begin migration and are located along the dorsal axis. Using an eyelash fixed to an inoculating stick (“hair knife”), a rectangular explant of neural crest tissue is isolated from the epidermis above and mesoderm below before removal from the embryo. Embryos from whic explants were dissected were either wild-type (some to be used as controls, some to be treated with chemical inhibitor) or injected at 4-cell stage with MMP-14 morpholino (20 ng). These explant were then plated onto fibronectin and allowed to bind to the surface. After two hours, experimental wild-type explants were treated with chemical inhibitor. Following complications with the solubility of NSC 405020 (which is soluble in DMSO and only sparingly soluble in MMR, in which the explants were incubated), only the chemical inhibitor Prinomastat hydrochloride was tested. This inhibitor disables the MMP-14’s sole activity and its ability to activate MMP-2; a concentration of 0.5 mM was tested. Explants were then imaged at a variety of times following treatment to observe migratory progress. Thus, within this experiment, three test groups were observed: wild-type control explants, explants from embryos that had been injection with 20ng MMP-14 morpholino (pre-translational knock-out), and explants treated with 0.5 mM of Prinomastat hydrochloride (post-translational knock-out).
The protocol for dissection of the explants is based upon the methods provided in Borchers et al. 2000. The embryos used for dissection are between the stages of 12.5 and 14, during early neurulation when neural crest cells begin migration and are located along the dorsal axis. Using an eyelash fixed to an inoculating stick (“hair knife”), a rectangular explant of neural crest tissue is isolated from the epidermis above and mesoderm below before removal from the embryo. Embryos from whic explants were dissected were either wild-type (some to be used as controls, some to be treated with chemical inhibitor) or injected at 4-cell stage with MMP-14 morpholino (20 ng). These explant were then plated onto fibronectin and allowed to bind to the surface. After two hours, experimental wild-type explants were treated with chemical inhibitor. Following complications with the solubility of NSC 405020 (which is soluble in DMSO and only sparingly soluble in MMR, in which the explants were incubated), only the chemical inhibitor Prinomastat hydrochloride was tested. This inhibitor disables the MMP-14’s sole activity and its ability to activate MMP-2; a concentration of 0.5 mM was tested. Explants were then imaged at a variety of times following treatment to observe migratory progress. Thus, within this experiment, three test groups were observed: wild-type control explants, explants from embryos that had been injection with 20ng MMP-14 morpholino (pre-translational knock-out), and explants treated with 0.5 mM of Prinomastat hydrochloride (post-translational knock-out).
Results:
Qualitative Analysis:
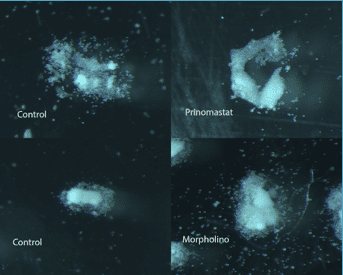
Explants in which the expression of MMP-14 was knocked-out experienced stunted migrations and, most significantly, individual cells did not separate effectively from each other and migrate individually. Instead, the tissue remained clumped and did not migrate in the diffuse way observed in the control explants. This result was observed more prominently in the chemically-inhibited explants and to a lesser degree in the morpholino-injected explants.
Quantitative Analysis:
The degree of migration will be quantified by cutting around the area in which the cells have dispersed and massing the pieces of the cut-out. Results have not yet been measured.
Conclusions:
These results deepen our understanding of the role of the MMP-14 protein in neural crest migration. Despite the ECM-digestive role of MMP-14 being controlled for in the use of the fibronectin plate, alteration of neural crest migration was observed in comparison to the control explants, particularly in chemically-inhibited explants. This suggests that MMP-14’s secondary roles in signaling pathways and activation of other matrix metalloproteinases may be significant in proper neural crest migration, possibly lending to a synergistic interplay between its primary and secondary functions. Further experimentation and additional repetition of this experiment is being conducted to increase our confidence and understanding of the result.
References:
Images:
https://classconnection.s3.amazonaws.com/223/flashcards/1190223/jpg/neurulation11334438927134.jpg
http://wiki.xenbase.org/xenwiki/index.php/Stage_14
Explants in which the expression of MMP-14 was knocked-out experienced stunted migrations and, most significantly, individual cells did not separate effectively from each other and migrate individually. Instead, the tissue remained clumped and did not migrate in the diffuse way observed in the control explants. This result was observed more prominently in the chemically-inhibited explants and to a lesser degree in the morpholino-injected explants.
Quantitative Analysis:
The degree of migration will be quantified by cutting around the area in which the cells have dispersed and massing the pieces of the cut-out. Results have not yet been measured.
Conclusions:
These results deepen our understanding of the role of the MMP-14 protein in neural crest migration. Despite the ECM-digestive role of MMP-14 being controlled for in the use of the fibronectin plate, alteration of neural crest migration was observed in comparison to the control explants, particularly in chemically-inhibited explants. This suggests that MMP-14’s secondary roles in signaling pathways and activation of other matrix metalloproteinases may be significant in proper neural crest migration, possibly lending to a synergistic interplay between its primary and secondary functions. Further experimentation and additional repetition of this experiment is being conducted to increase our confidence and understanding of the result.
References:
- Borchers A, Epperlein HH, Wedlich D. 2000. An assay system to study migratory behavior of cranial neural crest cells in Xenopus. Development Genes and Evolution 210: 217-222.
Images:
https://classconnection.s3.amazonaws.com/223/flashcards/1190223/jpg/neurulation11334438927134.jpg
http://wiki.xenbase.org/xenwiki/index.php/Stage_14
 RSS Feed
RSS Feed
